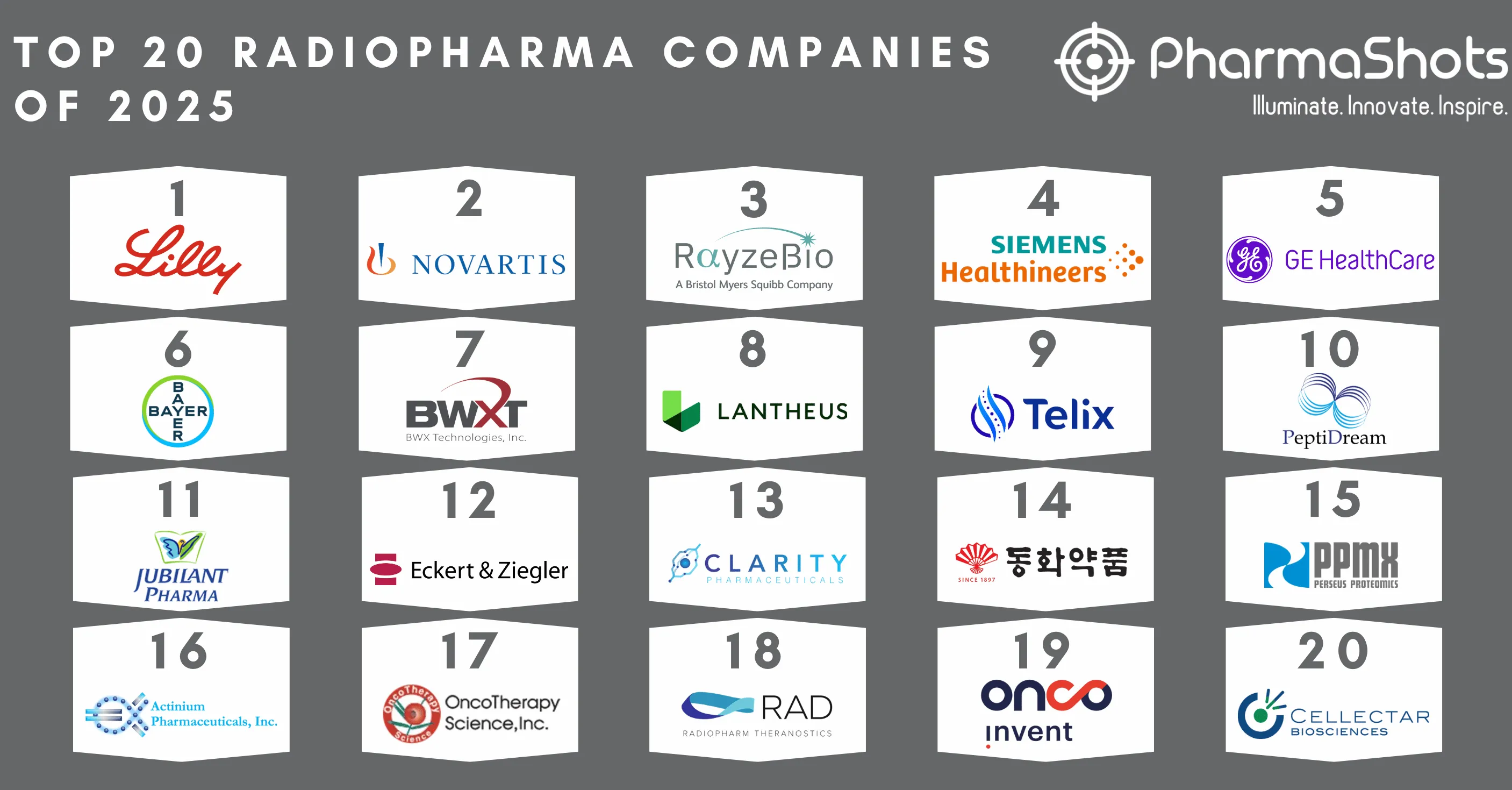
Top 20 Life Sciences Deals of 2022 by Total Deal Value
Shots:
- The technological advancement, shared cost, and the pursuit of combined extensive research are amongst the few pragmatic and mutual benefits that come with collaborations. Moreover, the risks involved in drug development plans get minimalized by opening the doors to new opportunities and commercialization
- When it comes to life science collaborations, companies seek beyond the traditional interests of the partnership. In 2022, Merck & Co. and Kelun-Biotech contributed to the largest life science deal with a total potential value of $9.47B followed by Sanofi’s deal with IGM Biosciences for up to $6.26B and Roche’s deal with Poseida Therapeutics for up to $6.22B
- With the invaluable insights of DealForma, PharmaShots has prepared this report on the Top 20 Deals in the Life Sciences industry of the year 2022 based on total announced deal value

Zymeworks' Development and Commercialization Deal with Jazz
Deal Date: Oct 19, 2022
Deal Value: $1.76B
Zymeworks granted Jazz the exclusive rights to develop and commercialize Zymeworks' Zanidatamab for the treatment of various indications across the US, EU, Japan, and other territories ex. Asia/Pacific regions. Under the terms of the agreement, Zymeworks received $50M up front and is also eligible for up to $525M in regulatory milestones and $862.5M in commercial milestones along with 10-20% of tiered royalties. Additionally, Zymeworks will also receive up to $325M as a one-time second payment if Jazz continues the collaboration following the clinical data from HERIZON-BTC-01. On Dec 21, 2022, Jazz exercised its option to resume the license agreement with Zymeworks to develop Zanidatamab.

Generate Biomedicines’ Development and Commercialization with Amgen
Deal Date: Jan 06, 2022
Deal Value: $1.90B
Generate granted Amgen the exclusive worldwide rights to develop and commercialize five clinical targets against multiple therapeutic areas and protein modalities along with an option to select up to 5 additional programs at additional costs. As per the terms of the transaction, Generate received $50M up front and is eligible to receive up to $1.85B or $370M as milestone payments for each program plus low double-digit royalties. If Amgen exercises its option for an additional program, Generate will receive additional undisclosed payments.

GentiBio’s Development and Commercialization Deal with BMS
Deal Date: Aug 10, 2022
Deal Value: $1.90B
GentiBio granted BMS the exclusive rights to develop and commercialize up to three Treg therapies. BMS will utilize GentiBio's T cell platform for the development of Treg therapies for Inflammatory Bowel Diseases (IBD). Under the agreemnet, GentiBio received an undisclosed upfront payment and is eligible for up to $1.9B in development, regulatory, and commercial milestones along with royalties.

Code Bio’s Research Collaboration with Takeda
Deal Date: Feb 22, 2022
Deal Value: $2.00B
Code Bio granted Takeda an exclusive license of four gene therapy programs. Code Bio and Takeda have agreed to develop gene therapies by leveraging Code Bio’s 3DNA non-viral genetic medicine delivery platform for rare diseases. Both companies will collaborate up to candidate selection process. Takeda will retain the responsibility for development and commercialization upon exercising the option. Moreover, Code Bio received a “double-digit million dollar” upfront payment and research funding. It is also eligible to receive future development and commercial milestones capped at $2B upon achievement of milestones for all four programs in addition to tiered royalties.

CytomX’s Development and Commercialization Deal with Regeneron
Deal Date: Nov 16, 2022
Deal Value: $2.03B
CytomX granted Regeneron the exclusive worldwide rights to develop and commercialize antibody-based immunotherapies. The collaboration combines CytomX's Probody platform and biologic masking expertise with Regeneron's Veloci-Bi bispecific antibody platform to develop immunotherapies for the treatment of cancer. Under the terms of the agreement, CytomX and Regeneron will jointly be responsible for the discovery of the antibodies whereas Regeneron will individually be responsible for funding preclinical, clinical, and commercial activities. Moreover, CytomX received $30M up front and is eligible for up to $2B in target nomination fees, development, regulatory, and commercial milestones, and single-digit to low-teen tiered royalties.

Jnana’s Development and Commercialization Deal with Roche
Deal Date: Nov 15, 2022
Deal Value: $2.05B
Jnana granted Roche the exclusive worldwide rights to develop and commercialize small molecule therapies for the treatment of cancer, immune-mediated and neurological diseases. These small molecule therapies will be developed using Jnana's RAPID chemoproteomics platform. As per the agreement, Jnana will be responsible for all the discovery and preclinical activities whereas Roche will be responsible for all clinical development and commercial activities. Jnana received $50M up front and is eligible for more than $2B in development, regulatory & commercial milestones along with tiered royalties.

Skyhawk’s Development and Commercialization Deal with Sanofi
Deal Date: Jul 05, 2022
Deal Value: $2.05B
Skyhawk granted Sanofi the exclusive worldwide rights to develop and commercialize small molecule therapies for the treatment of cancer and immunologic diseases by leveraging Skyhawk's SkySTAR platform. Under the terms of the agreement, Sanofi will be responsible for further development and commercialization following drug candidate status. Skyhawk received $54M up front and is eligible for up to $2B in development, regulatory, and commercial milestones, and royalties.

PeptiDream’s Development and Commercialization Deal with Merck & Co.
Deal Date: Dec 27, 2022
Deal Value: $2.1B
PeptiDream granted Merck the exclusive global rights to research, develop, and commercialize Peptide Drug Conjugate (PDC) therapies to treat multiple undisclosed targets. PeptiDream will provide Merck with the peptide candidates identified using its proprietary Peptide Discovery Platform System (PDPS) technology for use as PDCs against targets selected by Merck. Additionally, Merck will be responsible for the development and commercialization of these therapies. PeptiDream received an undisclosed upfront payment and is eligible for up to $2.1B in development, regulatory, and commercial milestones in addition to royalties.

POINT Biopharma’s Development and Commercialization Deal with Lantheus
Deal Date: Nov 11, 2022
Deal Value: $2.13B
POINT granted Lantheus the exclusive rights to co-develop and commercialize POINT's PNT2002 (PSMA-targeted 177 Lu-based radiopharmaceutical therapy) and PNT2003 (SSTR-targeted radioligand therapy) across the world except in certain Asian territories to treat mCRPC and GEP-NETs. Both PNT2002 and PNT2003 will be developed by combining POINT's radioligand development expertise with Lantheus' commercial expertise in PSMA PET and radiopharmaceuticals. As per the agreement. POINT will be responsible for the funding, development, and manufacturing of both products whereas Lantheus will be responsible for NDA approval along with development and commercial expenses. Additionally, POINT received $250M and $10M up front and is eligible for up to $281M and $34.5M in development milestones and up to $1.28B and $275M in commercial milestones including 20% and 15% royalties for PNT2002 and PNT2003 respectively.

Adagene’s Development and Commercialization Deal with Sanofi
Deal Date: Mar 02, 2022
Deal Value: $2.52B
Adagene granted Sanofi the right to develop and commercialize antibodies for cancer indications by using Adagene’s SAFEbody precision masking technology. Under the terms of the agreement, Adagene will be responsible for the early-stage research activities to develop masked versions of Sanofi candidate antibodies whereas Sanofi will be responsible for the late-stage research along with clinical, product development, and commercialization activities. Adagene retains the ability to advance Sanofi's two initial antibody candidates, followed by an option for two additional candidates. Adagene received $17.5M up front and is eligible for up to $2.5B in development, regulatory, and commercial milestones in addition to tiered royalties.

Century Therapeutics’ Research Partnership with Bristol Myers Squibb
Deal Date: Jan 10, 2022
Deal Value: $3.15B
Century Therapeutics entered into a research collaboration and license agreement with BMS to develop and commercialize up to 4 iPSC derived-engineered iNK and/or iT programs to treat hematologic malignancies and solid tumors. Century also granted BMS an option to license iPSC-derived allogeneic cell therapies to treat acute myeloid leukemia and multiple myeloma along with 2 additional programs. Century may elect to co-promote the AML program and one of the additional programs in the US for no exercise fee. Under the consent, Century received a $100M up front payment in cash and $50M in upfront equity investment at $23.14 per share. Century is eligible for up to $3B in development, regulatory, and commercial milestones across the four potential programs plus tiered high-single to low-double-digit royalties.

Wave Life’s Development and Commercialization Deal with GSK
Deal Date: Dec 13, 2022
Deal Value: $3.49B
Wave granted GSK the exclusive worldwide rights to develop and commercialize Wave’s WVE-006. The collaboration will combine Wave's PRISM platform with GSK's development and commercial capabilities to develop WVE-006 for the treatment of alpha-1 antitrypsin deficiency (AATD). Moreover, under a 4-year research collaboration, GSK will develop up to eight oligonucleotide therapies while Wave will develop up to 3 oligonucleotide therapies with an option to extend the research for an additional 3 years. As per the consent, Wave will lead all preclinical, regulatory, manufacturing, and clinical activities to Phase 1/2 trials for WVE-006 at their cost after which GSK will be responsible for registration and commercialization at their cost. Additionally, Wave will be responsible for preclinical activities for GSK and Wave therapies up to IND-enabling studies whereas both GSK and Wave will be responsible for the following development and commercialization of their respective programs. Wave received a $120M up front payment in cash and $50M in equity by granting 10683761 ordinary shares of Wave's common stock at $4.68 per share, and is eligible for up to $225M in development milestones and up to $300M in commercial milestones including tiered, double-digit to high teens royalties for WVE-006. Wave is also eligible for up to $150M in development milestones and up to $300M in commercial milestones including tiered, low-teens royalties for each GSK program. GSK is eligible for undisclosed commercial milestones and royalties for Wave programs

Orna’s Development and Commercialization Deal with Merck & Co.
Deal Date: Aug 16, 2022
Deal Value: $3.65B
Orna granted Merck rights to develop and commercialize multiple programs, including vaccines and therapeutics for cancer and infectious diseases indications. Orna's oRNA-LNP technology will be used to discover these products and Orna will retain rights to its oRNA-LNP technology. Under the terms of the agreement, Orna received $150M up front and is eligible for up to $3.5B in development, regulatory, and commercial milestones, plus royalties.

Arcellx’s Co-development and Co-commercialization Deal with Kite
Deal Date: Dec 09, 2022
Deal Value: $4.22B
Arcellx granted Kite the exclusive worldwide rights to develop and co-commercialize Arcellx's CAR T-cell therapy product designated as CART-ddBCMA, autologous CAR T-cell therapy designated as NextGen products, and non-autologous cell therapy designated as non-auto products for the treatment of myeloma. Kite also received an option to add two CAR T-cell therapy products including ACLX-001 and ARC-SparX products using Arcellx's ARC-SparX platform. Additionally, Arcellx has the option to co-develop and co-commercialize existing and NextGen products referred to as co-promoted products.
Arcellx will lead the P-II clinical trial (iMMagine-1) of CART-ddBCMA whereas Kite will develop the rest of the co-promoted products up to a prespecified term. Arcellx and Kite will be equally responsible for all additional expenses in the US and 60% (Kite) and 40% (Arcellx) ex. US. Kite will wholly be responsible for all non-co-promoted products and country-specific studies. Arcellx will be responsible for the manufacturing of the CART-ddBCMA up till its transfer after which Kite will be responsible for the manufacturing and all costs.
As per the transaction, Arcellx received a $225M up front payment in cash and $100M in equity by granting 3478261 shares of Arcellx's common stock to Gilead Sciences and is also eligible for up to $3.9B in clinical, regulatory, and commercial milestones. For co-promoted products, the companies will share cost and profit equally in the US and Arcellx is eligible for tiered, low to mid-teen royalties for other parts of the world. Whereas, for the non-co-promoted products, Arcellx is eligible to receive tiered, low to mid-teen royalties globally.

Arcturus’ Development and Commercialization Deal with CSL
Deal Date: Nov 01, 2022
Deal Value: $4.5B
Arcturus granted CSL’s Seqirus the exclusive global rights to develop and commercialize mRNA vaccines. Arcturus' self-amplifying mRNA (sa-mRNA) vaccine platform technology including STARR mRNA technology and LUNAR lipid-mediated delivery and other mRNA drug substance will be used for the development of these vaccines to treat COVID-19, influenza, and three other infectious diseases. Under the terms of the agreement, Arcturus received $200M up front and is eligible for up to $1.3B in development milestones and up to $3B in commercial milestones including low double-digit royalties for influenza and three other vaccines. Arcturus will also receive 40% of the profit from the COVID-19 vaccine sales.

Akeso’s Development and Commercialization Deal with Summit
Deal Date: Dec 05, 2022
Deal Value: $5B
Akeso granted Summit the exclusive rights to develop and commercialize the former’s PD-1/VEGF bispecific antibody, ivonescimab (AK112), to treat cancer including NSCLC in the US, Canada, Europe, and Japan. Akeso will retain rights to ivonescimab for the rest of the world including China and will be responsible for all R&D expenses including pre-clinical and clinical trials in non-licensed territories. Moreover, Summit will purchase certain drug substances for drug development. As per the consent, Akeso received a $500M up front payment and is eligible for up to $4.5B in development, regulatory, and commercial milestones in addition to low double-digit royalties.

Exscientia’s Development and Commercialization Deal with Sanofi
Deal Date: Jan 07, 2022
Deal Value: $5.3B
Exscientia granted Sanofi the exclusive global rights to develop and commercialize ~15 small molecule products under the therapeutic areas including oncology and immunology. Exscientia’s AI-driven platform will be used for the development of these small molecules. Exscientia will be responsible for drug design and lead optimization whereas Sanofi will be responsible for the preclinical and clinical development along with manufacturing and commercialization. As per the agreement, Exscientia received a $100M up front payment in cash and is eligible for up to $5.2B in research, translational, clinical development, regulatory, and commercial milestones in addition to tiered high-single-digit to mid-teens royalties. Exscientia also has an option for clinical co-investment to increase the royalty rate up to 21% for co-funded products.

Poseida’s Development and Commercialization Deal with Roche
Deal Date: Jul 30, 2022
Deal Value: $6.22B
Poseida granted Roche the exclusive worldwide rights for CAR-T cell therapy products including P-BCMA-ALLO1 & P-CD19CD20-ALLO1 (Tier 1 program) along with options for P-BCMACD19-ALLO1 & P-CD70-ALLO1 (Tier 2 program). Additionally, Roche also gained the rights to develop, manufacture & commercialize up to six CAR-T cell therapy products under the collaboration program and a non-exclusive right for up to three cell therapy products discovered by Roche for solid tumors. In accordance with the agreement, Poseida received $110M upfront, $110M in near-term payments & other payments along with eligibility to receive up to $6B milestone payments which includes $1.5B for the Tier 1 program, $1.1B for the Tier 2 program, $2.9B for collaboration programs & $415M for the licensed products. The company will also receive mid-single to low double-digit royalties for the Tier 1 and Tier 2 programs & low to mid-single digits royalties for licensed products.

IGM Biosciences’ Development and Commercialization Deal with Sanofi
Deal Date: Mar 29, 2022
Deal Value: $6.26B
IGM granted Sanofi the exclusive worldwide rights to develop and commercialize IgM antibody agonists to treat 3 oncology targets and 3 immuno/inflammation targets. For the oncology targets, IGM will be responsible for the R&D activities including related costs through the approval of the first BLA by the US FDA & EMA after which Sanofi will lead development and commercialization activities. The companies will jointly share 50:50 profits in certain major markets whereas IGM will be eligible to receive tiered royalties on global net sales. For immunology/inflammation targets, IGM will lead the R&D activities and related costs through the completion of the P-I clinical trial for up to 2 constructs whereas Sanofi will be responsible for further development and commercialization. IGM is eligible for tiered high single-digit to low-teens royalties globally. Under the consent, IGM received $150M up front and is eligible to receive up to $6.015B in regulatory and commercial milestones including $940M (for each oncology target) and $1.065B (for each immuno/inflammation target). In addition, Sanofi expressed an interest in purchasing up to $100M of IGM non-voting common stock in public financing.

Kelun-Biotech’s Development and Commercialization Deal with Merck
Deal Date: Dec 22, 2022
Deal Value: $9.47B
Kelun-Biotech granted Merck the exclusive global license to research, develop, manufacture, and commercialize seven preclinical ADC therapies for the treatment of cancers along with an option for additional ADCs. Kelun-Biotech held on to the rights to research, develop, manufacture, and commercialize certain licensed and option ADCs in China, Hong Kong, and Macau. Under the terms of the agreement, Kelun-Biotech received an upfront payment of $175M, an undisclosed equity investment, and is eligible for ~$9.3B in development, regulatory, and commercial milestones if the rights in China, Hong Kong, and Macau are not retained. Additionally, Kelun-Biotech is eligible to receive undisclosed tiered royalties.
Related Post: Top 20 Life Sciences Deals of 2021 by Total Deal Value
Tags

Shivani was a content writer at PharmaShots. She has a keen interest in recent innovations in the life sciences industry. She was covering news related to Product approvals, clinical trial results, and updates. We can be contacted at connect@pharmashots.com.